Cancer
In my Ph.D. thesis work, I investigated the structure/function relationship of the important enzyme poly(ADP-ribose) glycohydrolase (PARG) that hydrolyzes NAD-derived polymers of ADP-ribose (ADPR) that are formed by the poly(ADP-ribose) polymerase (PARP) family of enzymes. PARG is involved in the genotoxic stress response of the cell and its inhibition is thought to be beneficial in the treatment of some cancers.
My research led to the identification of a regulatory segment of PARG that is required for catalytic activity despite the fact that it is far removed linearly from catalytic residues [Botta, D. and Jacobson, M.K. (2010) Biochemistry. 49(35):7674-7682]. I then used structural data to make secondary structure predictions and to provide insight into the molecular composition of the different domains of PARG, whose 3-dimensional structure had not been determined at the time. I also worked collaboratively with the biotech company Trevigen Inc., in the optimization and validation of biochemical, high-throughput activity assays for PARP-1 and PARG [Botta, D. (2010) Thesis (Ph.D.). University of Arizona]. These assays were subsequently commercialized and have contributed significantly to the discovery of PARP-1 inhibitors as a new cancer therapy paradigm for the treatment of BRCA-deficient tumors.
My research led to the identification of a regulatory segment of PARG that is required for catalytic activity despite the fact that it is far removed linearly from catalytic residues [Botta, D. and Jacobson, M.K. (2010) Biochemistry. 49(35):7674-7682]. I then used structural data to make secondary structure predictions and to provide insight into the molecular composition of the different domains of PARG, whose 3-dimensional structure had not been determined at the time. I also worked collaboratively with the biotech company Trevigen Inc., in the optimization and validation of biochemical, high-throughput activity assays for PARP-1 and PARG [Botta, D. (2010) Thesis (Ph.D.). University of Arizona]. These assays were subsequently commercialized and have contributed significantly to the discovery of PARP-1 inhibitors as a new cancer therapy paradigm for the treatment of BRCA-deficient tumors.
|
My knowledge of NAD metabolism and expertise in assay development and optimization has also led to a fruitful collaboration with Southern Research, an incorporated affiliate of UAB, to identify small-molecule modulators of NAD(P) metabolism for the chemosensitization and treatment of hematological cancers.
|
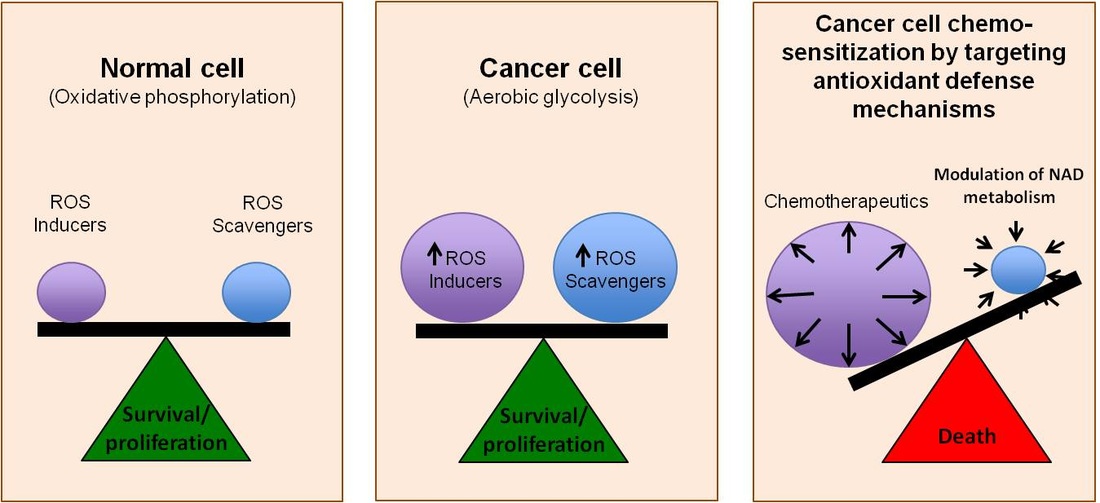
By means of a phenomenon termed “the Warburg effect,” tumor cells shift their energy production by mitochondrial oxidative phosphorylation to aerobic glycolysis, resulting in the upregulation of glucose consumption and increased production of reactive oxygen species (ROS). To compensate for the high levels of ROS, which can lead to cellular damage and death, cancer cells rely heavily on their antioxidant defense mechanisms. Figure 1 illustrates this phenomenon, which enables rapidly dividing cancer cells to survive despite the increased generation of ROS. The cellular antioxidant defense mechanism is largely controlled by the NAD(P)/NAD(P)H redox partners, and the purpose of this project, funded by the Alabama Drug Discovery Alliance, is to determine whether the modulation of NAD(P) metabolism can alter the cellular redox state, and whether its pharmacological modulation can be an effective anti-cancer therapeutic approach [Botta, D. et al. (2015) Cancer Res. 75(15 Suppl): Abstract nr 1242].